Single Technologies
Company
Single Technologies is based in Stockholm and was founded in 2012 [1]. Crunchbase lists them has having raised 4.7MUSD from JovB Holding, Sciety, and KTH Holdings [3]. There are ~10 employees on LinkedIn. The majority of the co-founders appear to come from a optical background.
Technology
Single have a few patents which largely refer to the optical system. The patents do not explicitly mention sequencing. What follows is based on one of their patents, and I’ll then try and frame this based on what they say on their website.
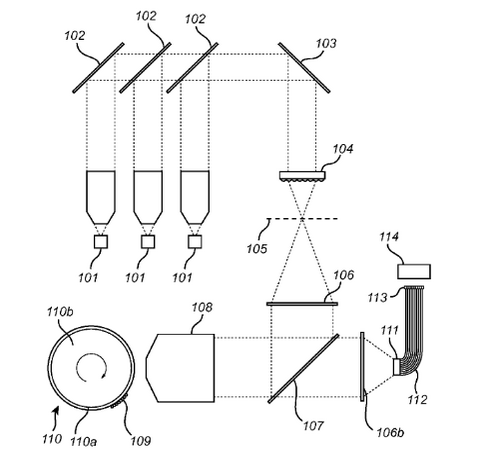
In The Single Technologies imaging system [2], the sample sits on a rotating sample holder. Essentially, it appears to be a drum that rotates under the objective lens. They suggest rotation means that the the sample is only subjected to constant forces. In a sense, this is similar to the TDI imaging that Illumina does on their instruments. The sample moves at a constant speed under the optical system and you essentially “scan” it. I can see that a curved sample surface is however a big departure from a traditional flat flowcell moving on an XY/XYZ stage.
The rotation trajectory appears to need to be very well defined, and they suggest using air bearings could help, and talk about precision (between laps) of 100nm. The patent describes the use of confocal microscopy, so rather than using a line scan imager (as in TDI) they will likely be scanning point-by-point. The instrument is called the “Theta” so it seems like a Confocal Theta Microscopy may also be a possibility.
From the patent we get the sense that they are innovating around the imaging system. The website more explicitly says that they are looking at single molecule detection. From the patent, and explicit mention of confocal microscopy on the site, I would not expect this imaging configuration to be compatible with “real-time” observation of nucleotide incorporation (PacBio-style).
The website mentions a patterned flowcell, but I didn’t see a patent referring to this. It would be interesting to better understand how this works, particularly in a single molecule context. One of the big issues with Illumina chemistry has been avoiding “mixed clusters”. These are clusters which are formed from more than one than template. Because the templates (DNA to be sequenced) randomly attached to the flowcell, there is some probability that two templates are very near each other and form a single “mixed” cluster. On the Genome Analyzer 2, these “mixed clusters” accounted for ~50% of data (from memory). Significantly limiting throughput.
If you have a number of wells/sites and are trying to optimize for “single occupancy”. Randomly flowing templates into wells limits you to ~36% [4] of wells containing a single template (with many containing none, or multiple templates).
Illumina solved this issue with their “exclusion amplification” chemistry [5]. In this approach as templates attach they are rapidly amplified quickly fill the well. This means that there isn’t any space left for a second template to enter a well. My understanding is that with this approach, most of the “mixed clusters” disappear, and you get a dramatic increase in reads.
Getting back to Single Technologies. My question is are you limited to only being able to use 36% of sites/wells? Or is there a single molecule approach to ensuring that each site only contains a single template? This would be quite interesting.
Outside of the optical system there doesn’t seem to be much to say about Single Technologies. They state that their approach “can be applied to almost any fluorescent based sequencing chemistry”. Which strongly suggests to me that they don’t have any innovation around chemistry. Looking at their team, I also don’t see employees with the background required to develop a new sequencing chemistry either.
So I suspect their plan is to innovate around the optics/flowcell only. Perhaps they can partner with someone else for the chemistry, or be acquired by an existing sequencing company [6], where they would provide a throughput advantage with improved optics/flowcells.
Notes
[1] https://www.singletechnologies.com/about-us
[2] http://www.freepatentsonline.com/y2019/0049382.html
[3] https://www.crunchbase.com/organization/single-technologies#section-funding-rounds
[4] https://books.google.co.jp/books?id=AJm7CwAAQBAJ&pg=PA43&lpg=PA43&dq=poisson+limit++single+occupancy+well&source=bl&ots=b33KVRG8fO&sig=ACfU3U2jGUbwzdGwzAqZ_LTHSiO5DZwJMg&hl=en&sa=X&ved=2ahUKEwj77dej68zpAhVVA4gKHcT0AJUQ6AEwCnoECAcQAQ#v=onepage&q=poisson%20limit%20%20single%20occupancy%20well&f=false
[5] https://www.illumina.com/science/technology/next-generation-sequencing/sequencing-technology/patterned-flow-cells.html
[6] I suspect of current players, only Illumina and BGIs chemistry would be compatible. In both cases this would mean removing amplification from their workflow so that they became single molecule approaches. The question then is if the Single Technologies approach can sufficiently reduce issues around photo bleaching to make this worth it. Moving to single molecule might help increase read length in some cases (by removing phasing issues) but if reduces average read length/throughput through photo bleaching, this might not be worth it.
Website Quotes
Left: DNA fragments located in SINGLE’s patterned flow cells. Middle: the Theta Sequencer. Right: sequencing data generated.
“The sequencer consists of the Worlds fastest scanner, a new type of fluidics adopted for large areas and sequencing chemistry.”
“Theta contains the world’s fastest single molecule-sensitive confocal imaging system”
“Single Technologies is pushing the limits of genomics by combining single molecule imaging, fast large area confocal scanning, grating techniques, fluidics, nanotechnology and a large portion of out of the box thinking. Our technology is being explored by leaders and industry in the genomics field who cares about Big Data generation.”
“Single Technologies was founded by Johan Strömqvist, Bengt Sahlgren, Annika Bolind Bågenholm and Raoul Stubbe in 2012/2013. The origin of the company is a unique combination of PhD research in single molecule imaging and biotechnology at the Royal Institute of Technology and R&D in the fiber optical grating industry by the founders of Proximion in Stockholm, Sweden.”
SCANNER
Theta contains the world’s fastest single molecule-sensitive confocal imaging system. The technology digitizes the samples simply and intuitively at diffraction limited resolution with negligible bleaching. And it’s ludicrously fast, a 15×15 mm area can be scanned in just a few seconds, and a total area of 125×65 mm could be scanned without any compromises.
FLUIDICS
Theta contains a new type of automated fluidics which avoids micro channels, allowing rapid exchange of liquids over large areas and using less reagents, setting new standards for optimized reactions. It is enabled by a combination of Single’s revolutionary scanning technology and unique approaches to effective flow and diffusion.
CHEMISTRY
Theta’s fast large area scanning and automated effective fluidics can be applied to almost any fluorescent based sequencing chemistry, supported by Single’s new patterned surface methods, giving the benefits of speed and capacity compared with other systems, backing the increased need of sequenced data.