BW Spectrometer Notes
- Meanwell PS45-5 40Watt 5V 8Amp open frame power supply
- Astrodyne OFM-0201 5V 4.4 Amp power supply
- Spectrometer control board
- B&W Tech fiber optic input spectrometer without TE cooling. Internal baffles will have to be removed to get the full spectrometer range, serial port output.
- OD6025-05HB 5V ball bearing fan.
- Keyswitch (key not included)
- Optical beamsplitter with assembly with lenses, beam splitter, fiber couplings
http://www.excaliburmineral.com/Aramis2/473nm/spectro/index.htm
http://www.excaliburmineral.com/Aramis2/473nm/index.htm
https://www.ebay.com/itm/162959836456?ViewItem=&item=162959836456
https://laserpointerforums.com/f42/b-wtech-473nm-unit-spectrometer-mods-info-101467.html
useful spectra: https://en.wikipedia.org/wiki/User:Deglr6328

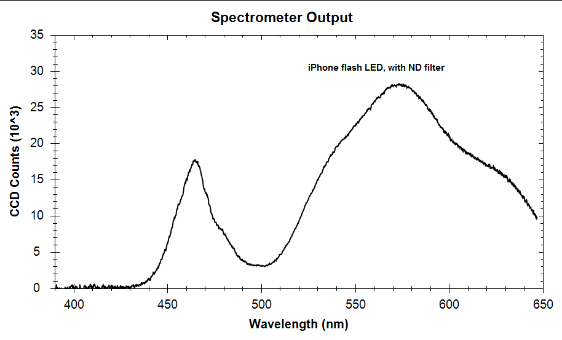
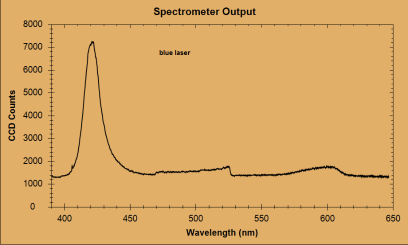
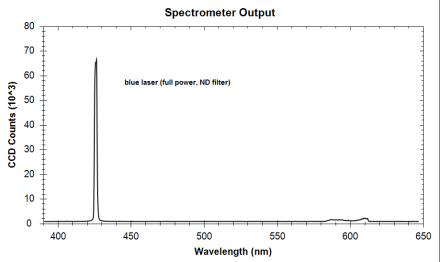
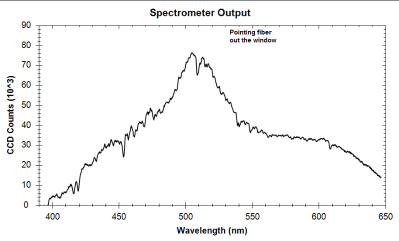




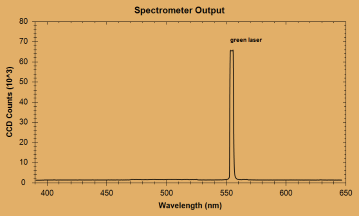
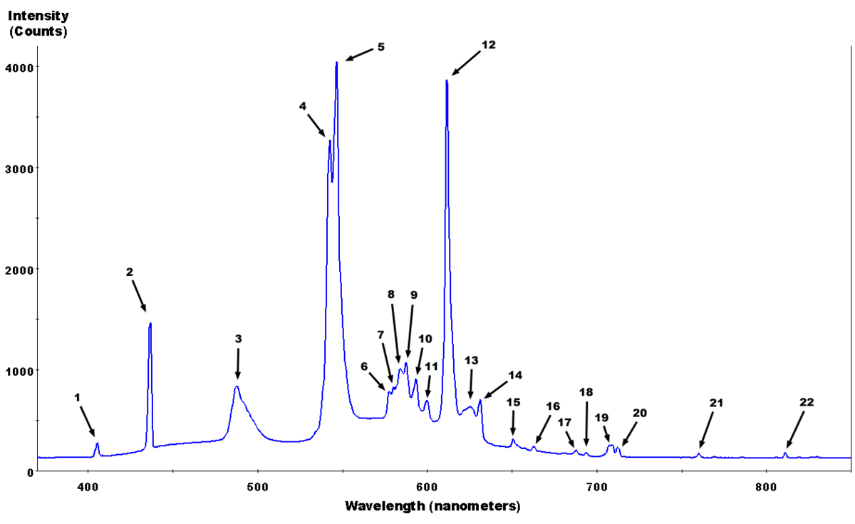
| Peak number | Wavelength of peak (nm) | Species producing peak | Actual line location (nm) |
|---|---|---|---|
| 1 | 405.4 | mercury | 404.656 |
| 2 | 436.6 | mercury | 435.833 |
| 3 | 487.7 | terbium from Tb3+ | ~485 to 490 |
| 4 | 542.4 | terbium from Tb3+ | ~543 to 544 |
| 5 | 546.5 | mercury | 546.074 |
| 6 | 577.7 | likely terbium from Tb3+ or mercury | 576.960 for Hg or ~578 for Tb |
| 7 | 580.2 | mercury or terbium from Tb3+ | 579.066 for Hg or ~580 for Tb |
| 8 | 584.0 | possibly terbium from Tb3+ or europium in Eu+3:Y2O3 | ~580 |
| 9 | 587.6 | likely europium in Eu+3:Y2O3 | ~587 |
| 10 | 593.4 | likely europium in Eu+3:Y2O3 | ~593 |
| 11 | 599.7 | likely europium in Eu+3:Y2O3 | ~598 |
| 12 | 611.6 | europium in Eu+3:Y2O3 | ~611 |
| 13 | 625.7 | likely terbium from Tb3+ | ~625 |
| 14 | 631.1 | likely europium in Eu+3:Y2O3 | ~630 |
| 15 | 650.8 | likely europium in Eu+3:Y2O3 | ~650 |
| 16 | 662.6 | likely europium in Eu+3:Y2O3 | ~661 |
| 17 | 687.7 | likely europium in Eu+3:Y2O3 | ~687-688 |
| 18 | 693.7 | likely europium in Eu+3:Y2O3 | ~693 |
| 19 | 707 and 709 | likely europium in Eu+3:Y2O3 | ~707 and ~709 |
| 20 | 712.3 | likely europium in Eu+3:Y2O3 | ~712 |
| 21 | 760.0 | likely argon | 758.9315 or 763.5106 (??) |
| 22 | 811.0 | likely argon | 811.531 |
General spectrometer info
Astro: http://www.threehillsobservatory.co.uk/astro/spectroscopy.htm
Hg Lines: http://www.astrosurf.com/buil/us/spe2/hresol4.htm
Hg Lines:
| Wavelength (nm) | Name (see photoresist) | Color |
|---|---|---|
| 184.45 | ultraviolet (UVC) | |
| 253.7 | ultraviolet (UVC) | |
| 365.4 | I-line | ultraviolet (UVA) |
| 404.7 | H-line | violet |
| 435.8 | G-line | blue |
| 546.1 | green | |
| 578.2 | yellow-orange |
- he Biophotonic Scanner is the world’s first measurement device to determine Carotenoid Value in the Skin (SCS) – an instant recording of carotenoid antioxidant activity in your body. By simply placing your hand in front of a faint blue light, you can get your Carotenoid Value in your skin within minutes. The Pharmanex® Biophotonic Scanner Technology is based on an optical method known as Raman Resonance Spectroscopy. This technology has been awarded the Nobel Prize and is used for biological measurements, as well as scientific discipline supported by years of research. Thanks to Pharmanex, technology is now available to everyone. The scanner uses optical signals to measure carotenoid levels in human tissue just below the skin’s surface. These signals identify the specific molecular structure of carotenoids. Pharmanex® is the exclusive holder of the patented Biophotonic Scanner technology. The scanner was developed by physicians and physicists at the top university in the United States
- Nava Whiteford, 2018/04/28 09:10http://www.thepulsar.be/article/science-surplus-spectrophotometer-review/
- ava Whiteford, 2018/04/28 13:38http://designengineusa.com/pharmanex/
- Nava Whiteford, 2018/04/28 15:52http://mmrc.caltech.edu/BWTec%20Examplar/Examplar.html